|
So that's why the number of electrons will also be 23 point no number of neutrons. So that's why the number of protons will be 23 and since it is a neutral element. The number of neutrons in an atom can be determined by the difference between the. So that will be equal to a number of electrons carrying negative charge, so that is equals to 23 point right. Therefore, a potassium atom has nineteen protons and nineteen electrons. 
Protons carry a positive electrical change, while electrons are negatively charged, and neutrons are neutral. Because potassium has 3 isotopes, then we have to calculate for the different atomic weights of the isotopes: Potassium-39 - It has 19 protons and atomic mass is 39. This missing mass is known as the mass defect, and you will learn about it in the chapter on nuclear chemistry. We can use the symbol, p, plus okay, because protons carry positive charge and since it is a neutral element, it do not carry any charge. Atoms are made of protons, neutrons, and electrons. (An observant student might notice that the sum of an atom’s subatomic particles does not equal the atom’s actual mass: The total mass of six protons, six neutrons, and six electrons is 12.0993 amu, slightly larger than 12.00 amu. So here atomic number that's equal to number of protons for proton. All atoms of potassium must have the same ( 1 ) atomic mass ( 2 ) atomic. Moving on to next part number 24 v, that is venarium. Atomic number is 19 and mass number is 41 point now. 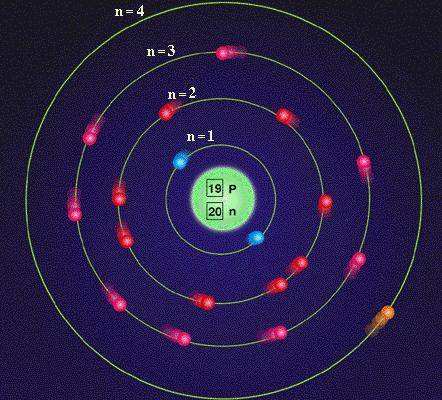
That is for potassium, we have atomic symbol here. Now also, we know that mass number, although we haven't asked for mass number, but we are solving it for lighting the symbol, so that's equal to number of proton plus neutron, so that is 19 plus 22 point that is 41 o, so atomic symbol. Basic Information Name: Potassium Symbol: K Atomic Number: 19 Atomic Mass: 39.0983 amu Melting Point: 63.65 C (336.8 K, 146.57 F) Boiling Point: 774.0 C (1047.15 K, 1425.2 F) Number of Protons/Electrons: 19 Number of Neutrons: 20 Classification: Alkali Metal Crystal Structure: Cubic Density 293 K: 0. It does not carry any charge, so this is further equals to number of electrons. Protons are a type of subatomic particle with a positive charge. 
The final subatomic particle was not discovered until 1932. Represent a single isotope of an element using the three main elemental symbolisms. We know that for potassium atomic number, that's equal to number of protons for potassium atomic number is 19 in number of protons will be 19 point. Electrons are a type of subatomic particle with a negative charge. Determine the number of protons, neutrons, and electrons in a specific isotope of an element.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
